Ĭhester FD (1891) Notes on three new or noteworthy diseases of plants. Int J Curr Microbiol App Sci 7:2632–2638. Crop Prot 90:150–156īhat ZA, Bhat MA, Ahanger MA, Badri ZA, Mir GH, Mohi-u-Din FA (2018) Survival of Didymella bryoniae incitant of ridge gourd blight under temperate conditions. Trop Agric 63:195–197īasım E, Basım H, Abdulai M, Baki D, Öztürk N (2016) Identification and characterization of Didymella bryoniae causing gummy stem blight disease of watermelon ( Citrullus lanatus) in Turkey. Plant Dis 99:1488–1499īala G, Hosein F (1986) Studies on gummy stem blight disease of cucurbits in Trinidad. 
īabu B, Kefialew YW, Li PF, Yang XP, George S, Newberry E, Paret ML (2015) Genetic characterization of Didymella bryoniae isolates infecting watermelon and other cucurbits in Florida and Georgia. 1 and 2).Īveskamp MM, de Gruyter J, Woudenberg JH, Verkley GJ, Crous PW (2010) Highlights of the Didymellaceae: a polyphasic approach to characterise Phoma and related pleosporalean genera. The accession number of the culture is NAIMCC-TF 2438 (Figs. The live culture has been deposited at National Agriculturally Important Microbial Culture Collection (NAIMCC), Mau, Uttar Pradesh, India, which is one of the culture collections registered by the World Federation for Culture Collections ( ). The sequence of the amplified product was deposited at NCBI with accession No. The amplicon obtained with this primer was of 735 bp size. Isolated DNA was also used to amplify with SCAR primers (RG-I-F 5’-TGTCGTTGACATCATTCCAGC-3′ and RG-I-R 5’-ACCACTCTGCTTAGTATCTGC-3′) which are specific to S. cucurbitacearum isolate (NCBI accession no.KC460840.1) available in the database submitted by Liu et al. 
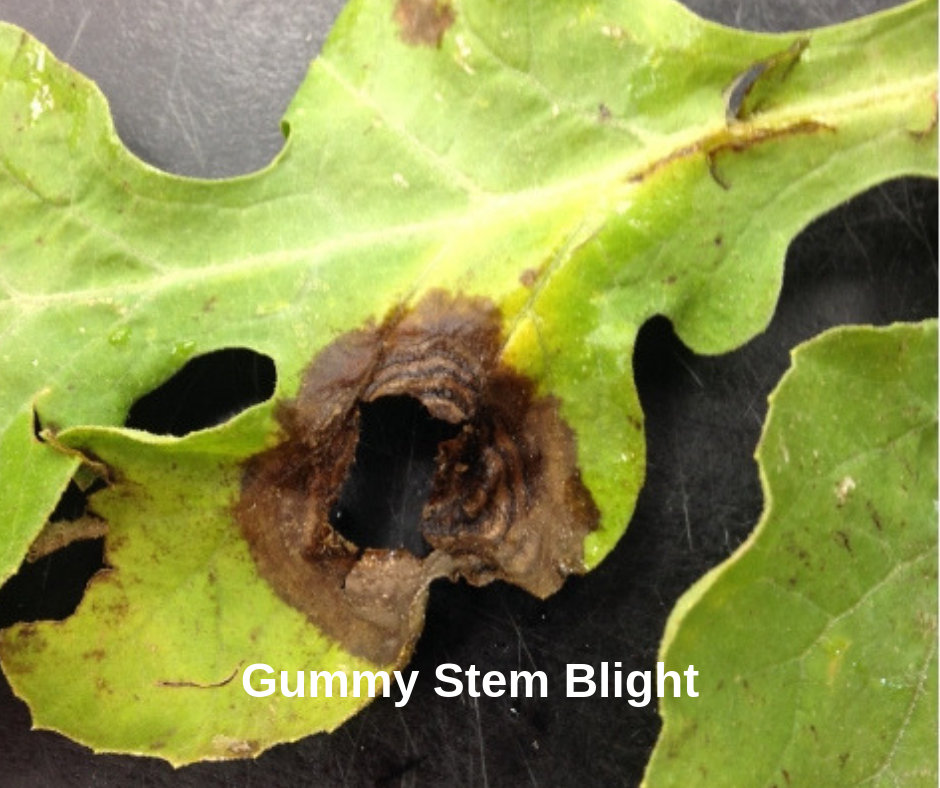
The sequence of the isolate was compared with the NCBI – GenBank database using the blast algorithm ( Results confirmed that the fungal isolate was sharing 99% homology with the S. Remaining PCR product was purified and sequencing was performed in ABI-3710 Prison automated DNA analyser (Eurofins, India). The ethidium bromide stained agarose gel was visualised under UV light (UVITEC FIREREADER V.10, Cambridge, U.K). PCR product of 5 μL volume was loaded on to 1% ( w/ v) agarose gel and electrophoresed at 75 V for 1.5 h. PCR conditions included an initial denaturation step at 94 ☌ for 2 min followed by 35 cycles of denaturation at 94 ☌ for 30 s., annealing at 52 ☌ for 30 s., extension at 72 ☌ for 30 s., followed by a final extension at 72 ☌ for 5 min. Taq (GeNei, India), and one μL DNA template. Reaction components consisted of 2.5 μL of 10X PCR buffer with 25 mM MgCl 2 (GeNei, India), 0.5 μL10 mM dNTPs (GeNei, India), 0.5 μL of10 μM forward and reverse primers, 0.3 μL 3 U/μL. 2014) and PCR reactions were carried out in a total volume of 25 μL. Genomic DNA was extracted by CTAB method (Mishra et al. The fungus has been characterised molecularly using ITS primers viz., ITS 1 and ITS 4 (White et al. Initially mycelium was bright white in colour later turning into light olive-green colour as reported by Keinath et al. A thick layer of mycelium appeared just seven days after transfer without any sporulation. After two days when growth was observed on plates, the growing tip of the fungal colony was transferred to another freshly prepared PDA plate for purification. The dried pieces of infected stems were placed on potato dextrose agar (PDA) and kept incubated at ambient conditions. Samples were kept on pre-sterilised blotting paper for 5 min for air-drying. These bits of 5 mm length and 2 mm width were washed in normal water followed by surface sterilisation with 0.1% NaOCl for 2 min and rinsed twice with sterile distilled water to make it free from any surface contamination. Samples were collected from the infected watermelon plant from the symptomatic region of stem. We attempted to isolate and characterise the gummy stem blight pathogen of watermelon at the Indian Institute of Horticultural Research, Bengaluru, India from diseased plants observed in our research farm. It causes complete wilting of plant with an estimated yield loss up to 100% (Bala and Hosein 1986). In severe cases, the pathogen enters into the vascular bundles of plant and clogs the phloem tissue and later damaging the xylem. Later, the pathogen spreads to cover complete leaf, turning it dark brown and dry affecting photosynthesis and yield. Disease symptoms initially start with foliar marginal brown discolouration, spreading towards the inside leaf lamina. Recently, GSB has become a threat for the production of watermelon that has not been reported earlier in this region. Watermelon ( Citrullus lanatus) is an important commercial, cucurbitaceous vegetable cultivated throughout the year in Bengaluru, India. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
